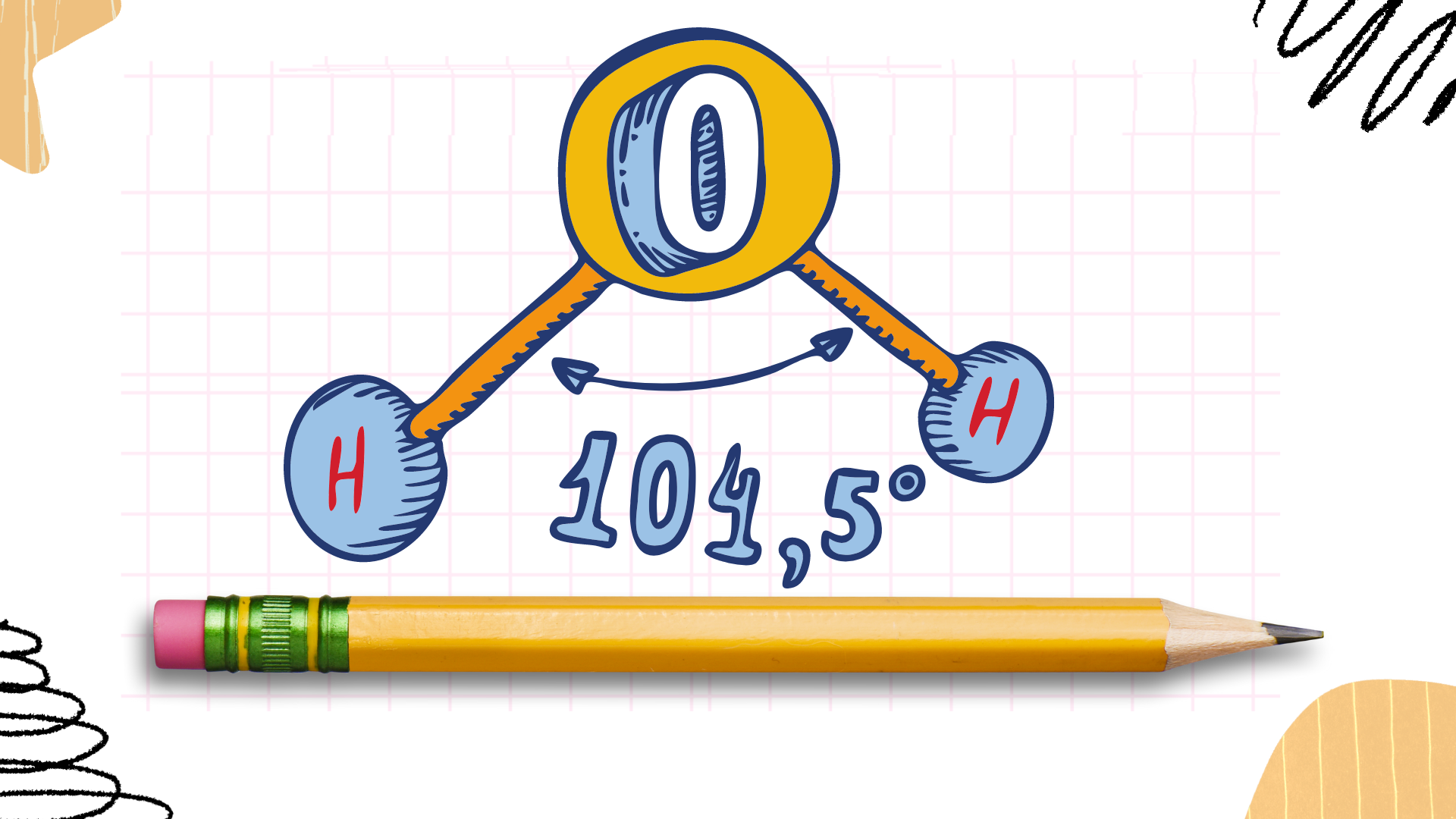
The Lewis dot structure is a simple yet powerful representation of the electron arrangement in a molecule. In this article, we will explore the Lewis dot structure of water (H2O) and delve into the underlying molecular bonding that gives water its unique properties.
Understanding Lewis Dot Structures
Lewis dot structures, also known as electron dot structures or Lewis structures, provide a visual representation of the valence electrons in an atom. Valence electrons are the electrons in the outermost energy level of an atom and play a crucial role in determining an element’s chemical behavior and bonding capabilities.
In a Lewis dot structure, each valence electron is represented as a dot placed around the symbol of the element. These dots are distributed in pairs, usually placed on the four sides of the element’s symbol.
The Lewis Dot Structure of H2O
Water, with the chemical formula H2O, consists of two hydrogen (H) atoms and one oxygen (O) atom. To create the Lewis dot structure for water, we follow these steps:
- Determine the Total Number of Valence Electrons: Oxygen has 6 valence electrons (located in the 2s and 2p orbitals), while hydrogen has 1 valence electron each. Therefore, for H2O, we have a total of 6 + 2(1) = 8 valence electrons.
- Place the Atoms in the Structure: Oxygen is the central atom in the H2O molecule, as it is more electronegative than hydrogen and can accommodate more electrons around it. Hydrogen atoms are placed on either side of the oxygen atom.
- Distribute Valence Electrons: Start by placing lone pairs (pairs of valence electrons) around the oxygen atom to satisfy its octet rule (oxygen wants 8 electrons around it). In this case, two pairs of electrons will be placed around oxygen.
O
/ \
H H- Distribute Remaining Electrons: After accounting for the lone pairs, distribute the remaining electrons around the hydrogen atoms. Each hydrogen atom can share one electron with oxygen, forming a single covalent bond.
H H
| |
OUnderstanding Molecular Bonding in H2O
The Lewis dot structure of water reveals that each hydrogen atom forms a single covalent bond with the central oxygen atom. A covalent bond is formed when atoms share electrons to achieve a more stable electron configuration. In the case of water, oxygen shares one electron with each hydrogen, resulting in the sharing of a total of two electrons.
This sharing of electrons creates a polar covalent bond between the hydrogen and oxygen atoms. Oxygen’s higher electronegativity attracts the shared electrons more strongly, causing a partial negative charge (δ-) on the oxygen atom and partial positive charges (δ+) on the hydrogen atoms. This polarity gives rise to water’s unique properties, such as its ability to form hydrogen bonds and exhibit strong cohesive and adhesive forces.
The Lewis dot structure of H2O provides insight into the arrangement of valence electrons and the molecular bonding that underlies the properties of water. This simple representation helps us understand how the polar covalent bonds in water give rise to its remarkable characteristics, making it an essential substance for life and a key player in various chemical and biological processes.